 Here, H represents hybridization on the central atom, V as valence electrons on the central atom of the molecule, M as the monovalent atoms bonded to the central atom, Cas charge on cation or more electropositive atom, and A represents the charge on anion or more electropositive atom. The formula to find the Hybridization of any molecule is given below:. So when we add the number of bonded sites and lone pairs we get the value of H as 2+3 = 5, which means that IF3 is Sp3d hybridized. It is bonded to 3 atoms of F and also has 2 lone pairs on it. We know that the Iodine atom is the central atom in IF3. If H=2 denotes sp hybridization, H=3 denotes sp2 hybridization, H=4 denotes sp3 hybridization, H=5 denotes sp3d hybridization, and H=6 means sp3d2 hybridization. The value of Hybridization (H) is determined by:. Hybridization of any molecule can be found by adding the total number of bonds formed by the central atom and the number of lone pairs on the central atom. Hybridization of any molecule can found using the given methods:. Hybridization helps us to understand more about the nature of bonding in a molecule and is useful in determining the best, stable molecular shape of a molecule.Īs the name suggests, molecules form a combination of hybrid orbitals to achieve more stability. Thus, this Lewis structure of IF3 with 2 lone pairs is the most stable with each atom having a formal charge of 0.  
Hence we do not need to convert the lone pairs into double/ triple bonds since we already have the best Lewis structure of the molecule IF3. If we check the formal charge of each atom for the molecule we see that each atom has a value of 0 which is the lowest possible. Now that we have our Lewis structure, let us make sure that it is the best Lewis structure possible for IF3. Hence I have 2 lone pairs and can hold more than 8 valence electrons. It can hold more than 8 valence electrons because atoms below period 5 can have an expanded octet and thus can carry more valence electrons. Now I have more than 8 valence electrons. So, these valence electrons will act as lone pairs on the central atom I. We still have 4 valence electrons remaining. Now we arrange the valence electrons around each atom so that it completes its octet.Īfter completing the above procedure, we see that 24 valence electrons are used up. Since there are three atoms of F, 6 valence electrons are used up. Now we start arranging the electrons as a chemical bond which is represented by two dots. So by definition, the central atom in IF3 is I. The atom having the highest valence and the higher number of bonding sites is considered the central atom. Hence the total valence electrons for the molecule IF3 counts to 21+7= 28. But there are 3 atoms of F, so it will have 7×3= 21 valence electrons. I and F both belong to group 7 and hence have 7 valence electrons each. Let us start by counting the total number of valence electrons for the molecule IF3. The formal charge can be calculated by using the formula:. 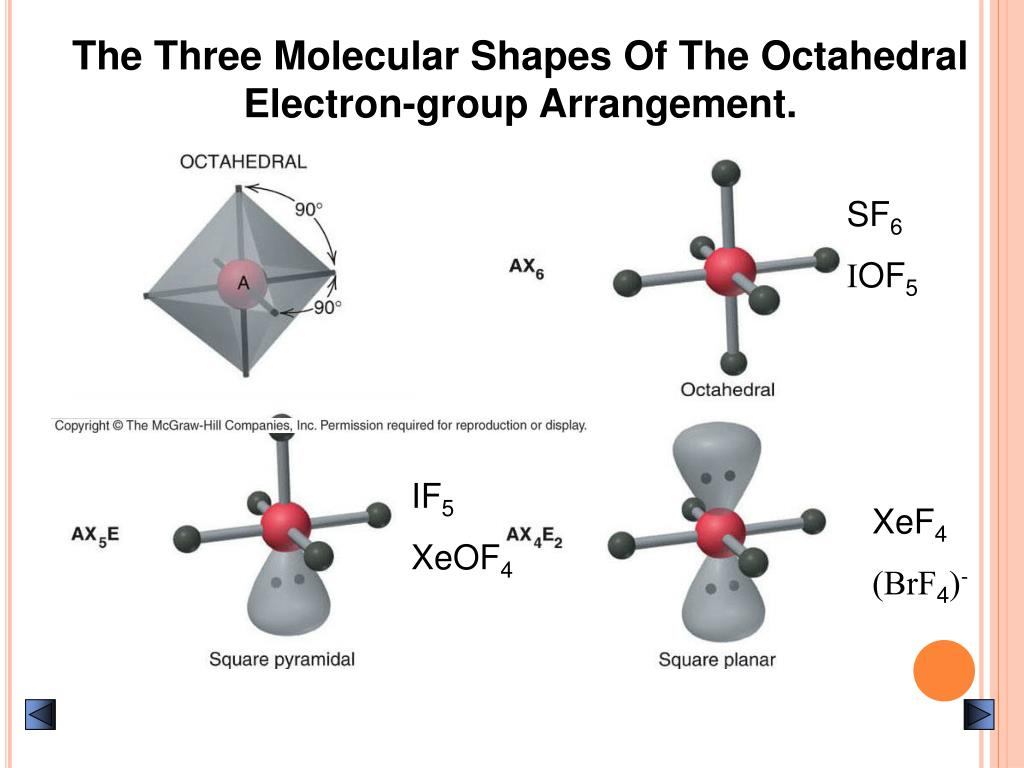
The best Lewis structure for any molecule should have each atom with a formal charge of 0. If possible, convert lone pairs into double or triple bonds depending on the stability of the molecule.Īs a last resort, we should always check the formal charge of each atom and make sure that it is the lowest possible. Now we fill in the remaining valence electrons so that each atom has completed its octet which makes the overall molecule stable. Now we start drawing chemical bonds between the central atom and the surrounding atoms. Next, we need to identify the central atom of the molecule. The first and foremost step is to count the total number of valence electrons of the molecule. Let us check out the steps one should follow for drawing the Lewis structure:. The Lewis structure of IF3 shows that I is surrounded by 2 lone pairs of electrons and forms 3 single bonds with each of the F atoms. As you already know, valence electrons are used to draw the Lewis structures of any molecule. Before we begin to learn the steps for drawing Lewis structure let’s see the Lewis structure of IF3. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
